In the search for more, new and cleaner sources of energy, a largely untapped resource is emerging: natural hydrogen.
Unlike hydrogen produced from industrial processes, natural hydrogen forms through geological reactions that occur normally within the Earth’s crust, meaning it costs nothing to make – though it costs some amount to extract – and does not emit any carbon dioxide or other human‑caused pollutants.
Today, hydrogen is used mainly in oil refining, production of ammonia for fertilizer and to make methanol, which can be a fuel and an ingredient in plastics. Emerging technologies are making hydrogen a viable fuel for cars, planes, ships and factories. Hydrogen demand around the world is projected to grow from around 90 million metric tons in 2022 to more than 500 million metric tons by 2050. Some of that supply could come from nature itself, as well.
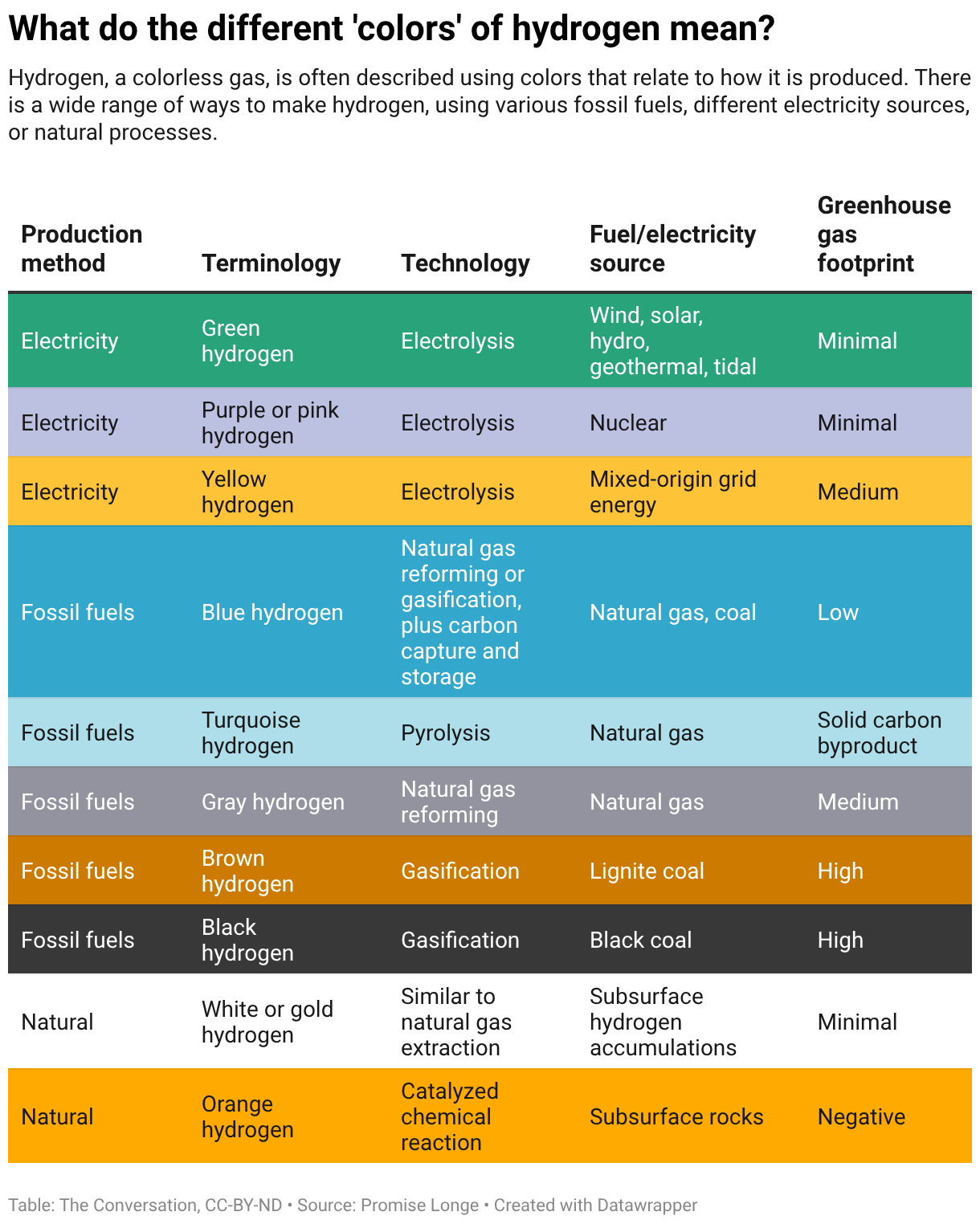
To describe each source of hydrogen, energy researchers like me, and the energy industry as a whole, use a range of colors. In general, “gray” and “blue” hydrogen are made by burning fossil fuels, with blue hydrogen incorporating technology that captures the carbon dioxide produced in the process to reduce emissions. “Green” hydrogen comes from renewable‑energy‑powered electrolysis, using electricity to split water into hydrogen and oxygen. “White” or “gold” hydrogen occurs naturally underground and can be extracted directly with minimal processing.
How natural hydrogen forms
Natural hydrogen originates from several geological processes. The most well‑studied mechanism is serpentinization, a reaction where water interacts with iron‑rich rocks known as ultramafics, releasing hydrogen gas.
Serpentinization occurs in diverse settings around the world, including ocean ridges and continental formations such as the Midcontinent Rift in North America, a band of mostly igneous rocks with some sedimentary rocks mixed in, which extends from Minnesota through the Lake Superior region and southward toward Kansas.
Another process, thermogenic hydrogen formation, occurs in deep sedimentary basins when organic material decomposes under high temperatures, roughly 480 to 930 degrees Fahrenheit (250 to 500 degrees Celsius). These reactions can also produce hydrogen alongside other gases, such as methane or nitrogen.
Because these processes happen over millions of years, using natural hydrogen generally requires far less energy than human‑made methods such as electrolysis, which consumes roughly 50 kilowatt-hours of electricity per kilogram of hydrogen produced – enough to power an average home for a day or two, and more than the energy that kilogram of hydrogen can provide. Natural hydrogen is already made – it just has to be collected.
The science and the search
Researchers and exploration companies are developing methods similar to those used in oil and gas exploration to locate potential hydrogen accumulations. They are looking at three types of geological formations:
Focused seepage, where hydrogen seeps naturally through cracks and faults. It tends to reach the surface and disperse quickly, making large-scale capture difficult.
Coal beds, where hydrogen binds to coal layers, offer higher potential density but pose difficulties for extraction. The hydrogen must first be separated from the coal and then flow through tight rock layers to the extraction point.
Reservoir‑trap‑seal systems, comparable to the rock formations that trap natural gas underground, are considered the most promising for commercial production because they can concentrate large volumes of hydrogen in well‑defined, drillable structures. However, they remain largely unproven in practice: The basic idea is well established, and geologists have a good sense of where those formations might occur, but they still lack detailed data on how much hydrogen these formations actually contain and how easy it would be to extract.

Massive reserves – somewhere
The U.S. Geological Survey estimates there could be more than 5 trillion metric tons of geological hydrogen underground around the world. But only a small fraction of that is estimated to be recoverable, both technically and in terms of reasonable costs.
However, even 2% of that total would be more than all proven natural gas reserves on the planetand enough to meet projected demand for the next 200 years, even accounting for increased consumption.
All of that reserve has built up over billions of years. The Earth naturally produces between 15 million and 31 million metric tons of natural hydrogen each year – less than 1% of the amount expected to be needed each year by 2050. But only a fraction of that is likely to be efficiently captured.
So geologic hydrogen is likely best viewed as a very large but ultimately finite source of low‑carbon energy that can substantially complement, but not replace, other energy sources, including various methods of producing hydrogen.
Global hot spots
Currently, only one hydrogen field, at Mali’s Bourakébougou village, produces natural hydrogen commercially, supplying tens of tons of hydrogen per year to power the village.
However, the number of companies exploring for natural hydrogen has increased rapidly, from roughly 10 in 2020 to about 40 by the end of 2023, according to Rystad Energy and related government and research‑lab reports.
Apart from that one field in Mali, exploration is concentrated in the United States, Australia, Canada and several European countries.
In the U.S., HyTerra’s Nemaha Project in Kansas has confirmed subsurface hydrogen concentrations reaching more than 90% hydrogen and 3% helium. The higher the concentration of hydrogen, the more efficient and cost‑effective it is to recover. HyTerra is also exploring elsewhere in the Midwest and Rocky Mountain regions.

Technical barriers
Transforming geological hydrogen into a commercial energy source presents tough scientific and technical challenges. Detecting and measuring hydrogen underground is difficult because of its small molecular size and reactivity with other elements in the rocks.
And if what’s found is low concentrations of hydrogen mixed with large amounts of other gases, it can be costly, even prohibitively so, to separate and purify the hydrogen before it can be used.
Economics and efficiency
The economic promise of natural hydrogen lies in its simplicity.
Because geological processes already performed the production work, early estimates suggest that extraction costs could be one‑tenth the production costs for other traditional hydrogen generation techniques – or possibly even less than that.
But those figures are based on the small amounts of hydrogen found so far and may not represent future large‑scale performance. Producing enough to serve commercial demand will require discovering large, high-quality accumulations.
As one leading research group noted, “This is not a gold rush.” It’s a careful exploration for scientific evidence that could lead, in time, to an abundant, carbon‑free and continuous energy source that complements other renewable energy sources.
This article is republished from The Conversation, a nonprofit, independent news organization bringing you facts and trustworthy analysis to help you make sense of our complex world. It was written by: Promise Longe, University of Kansas
Read more:
- What is hydrogen, and can it really become a climate change solution?
- How the ‘Big Beautiful Bill’ positions US energy to be more costly for consumers and the climate
- Lightweight of periodic table plays big role in life on Earth
Promise Longe does not work for, consult, own shares in or receive funding from any company or organization that would benefit from this article, and has disclosed no relevant affiliations beyond their academic appointment.






















(0) comments
Welcome to the discussion.
Log In
Keep it Clean. Please avoid obscene, vulgar, lewd, racist or sexually-oriented language.
PLEASE TURN OFF YOUR CAPS LOCK.
Don't Threaten. Threats of harming another person will not be tolerated.
Be Truthful. Don't knowingly lie about anyone or anything.
Be Nice. No racism, sexism or any sort of -ism that is degrading to another person.
Be Proactive. Use the 'Report' link on each comment to let us know of abusive posts.
Share with Us. We'd love to hear eyewitness accounts, the history behind an article.